TiARABAT
TiO2-based Anodes for Rechargeable Aqueous Proton BATteries
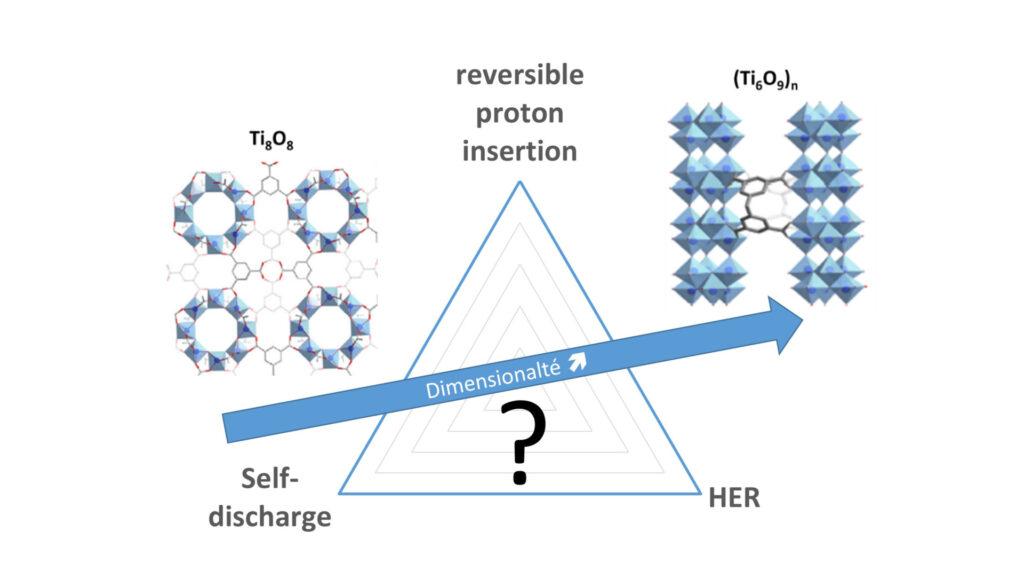
TiO2-based materials have been recently identified as suitable anode materials for the development of large-scale eco-sustainable aqueous batteries (with applications for the storage of electricity produced by renewable energies at the grid level), notably when relying on fast, massive and reversible proton insertion. However, parasitic reactions such as hydrogen evolution and self-discharge currently limits their performances. The purpose of the TiARABAT projet is to provide a comprehensive understanding on the proton-coupled electron transfer reactions occurring at TiO2-based electrodes, notably by taking advantage of new materials such as the versatile Titanium based Metal-Organic Frameworks, in which the size/structure of the Titanium oxide core can be finely tuned (ranging from 1D nanowires to discrete clusters). By implementing a rigorous electroanalytical methodology and associating an experimental approach relying on aqueous electrolytes of various composition/pH with multiphysics simulations, we will deconvolute and rationalize the contribution of each process to the electrochemical reactivity of a range of composite electrodes differing by the structure of the Titanium oxide core. These new results, combined with previous one obtained at bulk TiO2 electrodes, will allow to establish the structure-function relationship of TiO2-based materials, a prerequisite for the rational design of structures optimized for reversible proton insertion.